
The goal of these experiments was to establish the feasibility of human stem cell engraftment in the mouse cochlea in vivo. 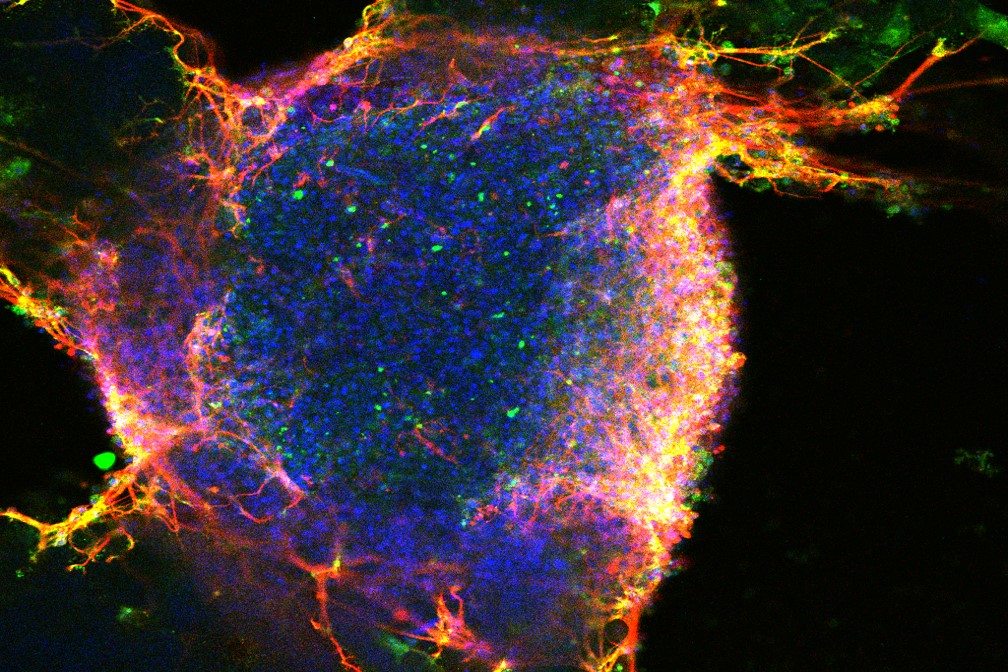
Using this model as well as wild type (WT) controls, we assess the survival and engraftment of transplanted human pluripotent stem cell-derived otic progenitors. To do this, we used Pou4f3DTR/+, an established mouse model for acute cochlear hair cell loss to induce hair cell loss with diphtheria toxin injections. Furthermore, as Lang and colleagues have demonstrated that the ablated sensory epithelium may be more amenable to the migration and engraftment of transplanted cells, we sought to use an acutely deafened model in our studies. 
Based on the existing literature on the successful engraftment at an early age in the chick inner ear, we used neonatal mice as transplantation recipients. We used a round window (RW) injection into the scala tympani perilymph as our route of delivery of the cells to increase the chances of cell survival (avoiding high K+ environment of the scala media endolymph) and migration into the OC through the basilar membrane. We focus our investigations on transplanting stem cells at an early otic fate, and therefore, we differentiated human embryonic stem cells (hESC) to pre-placodal ectoderm (PPE) using previously reported methods. Here we test the plausibility of grafting human pluripotent stem cell-derived otic progenitors into a murine cochlea after induced hair cell loss. Multiple investigators have reported successful engraftment of mouse stem cells in the mouse, guinea pig, and rat OC, with the consistent finding of only a small number of cells engrafting successfully. However to date, little is known of the ability of human cells to engraft in the mammalian auditory sensory epithelium, the organ of Corti (OC). Other xenograft models combining an avian recipient of mammalian stem cells showed successful engraftment and differentiation of mouse stem cell-derived progenitor cells into the nascent chick auditory sensory epithelium. Furthermore, human neuronal progenitor cells have been successfully engrafted into the injured gerbil modiolus leading to restoration of hearing. Successful generation of otic progenitor and inner ear hair cells from mouse or human stem cells has been elegantly demonstrated in vitro. One strategy for stem cell-based therapies involves in vitro generation of the inner ear (otic) progenitor cells followed by their subsequent transplantation into the deafened cochlea (in-depth review ). The use of pluripotent stem cells for this purpose represents one potential promising such method. Therefore, the development of the methods for hair cell regeneration or replacement would help in establishing curative therapies for sensory hearing loss in the future. Furthermore, mammalian hearing loss is irreversible, because mammalian HCs, unlike those of birds, do not regenerate spontaneously. Hearing loss is most commonly caused by inner ear cochlear hair cell (HC) death, and the resulting hearing deficits have a profound, negative impact on the daily life of affected people. We have demonstrated the feasibility of human stem cell engraftment into an ablated mouse organ of Corti. We demonstrate that hair cell ablation prior to transplantation leads to increased engraftment in the auditory sensory epithelium, the organ of Corti, as well as differentiation of transplanted cells into hair and supporting cell immunophenotypes. We differentiate human pluripotent stem cells to the pre-placodal stage in vitro then transplant them into the mouse cochlea after selective and complete lesioning of the endogenous population of hair cells. However, the ability of human stem cells to engraft and generate differentiated cell types in the inner ear is not well understood. A number of investigators have previously demonstrated successful implantation, and certain level of regeneration of hair and supporting cells in both avian and mammalian models using rodent pluripotent stem cells. Stem cell-based therapies are one such strategy and offer promise for cell replacement in a variety of tissues. As a result, there is a need for the development of regenerative strategies to replace hair cells once lost. Loss of the hair cells of the inner ear commonly underlies deafness and once lost this cell type cannot spontaneously regenerate in higher vertebrates. 
Hearing loss affects 25% of the population at ages 60–69 years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
